Introduction
This vignette covers the first step in the xcms metabolomics data analysis workflow: visualizing raw MS data before any processing. These visualizations help you:
- Assess data quality before analysis
- Understand the structure of your LC-MS acquisition
- Identify potential issues early
- Visualize MS/MS (DDA) experiment coverage
xcms Workflow Context
┌───────────────────────────────┐
│ 1. RAW DATA VISUALIZATION | ← YOU ARE HERE
├────────────────────────────-──┤
│ 2. Peak Detection │
│ 3. Peak Correspondence │
│ 4. Retention Time Alignment │
│ 5. Feature Grouping │
└──────────────────────────----─┘Functions Covered
| Function | Purpose | Input Type |
|---|---|---|
gplot(XcmsExperiment) |
Visualize full MS data |
XcmsExperiment, XCMSnExp
|
gplotPrecursorIons() |
Visualize MS/MS precursors |
MsExperiment with MS2 |
Setup
Part 1: Full MS Data Visualization
Overview
The gplot() method for XcmsExperiment and XCMSnExp objects creates a two-panel visualization:
- Upper panel: Base Peak Intensity (BPI) chromatogram vs retention time
- Lower panel: m/z vs retention time scatter plot
Both panels use intensity-based coloring, and detected peaks (if any) are automatically overlaid as rectangles.
Data Preparation
We’ll use pre-processed test data from xcms:
# Load pre-processed data
xdata <- loadXcmsData("faahko_sub2")
# Check data
cat("Samples:", length(xdata), "\n")
#> Samples: 3
cat("Total peaks detected:", nrow(chromPeaks(xdata)), "\n")
#> Total peaks detected: 248For visualization, we’ll filter to a specific retention time and m/z region:
# Filter to focused region
mse <- filterRt(xdata, rt = c(2785-100, 2785+100))
mse <- filterMzRange(mse, mz = c(278, 283))Basic Usage
Single Sample Visualization
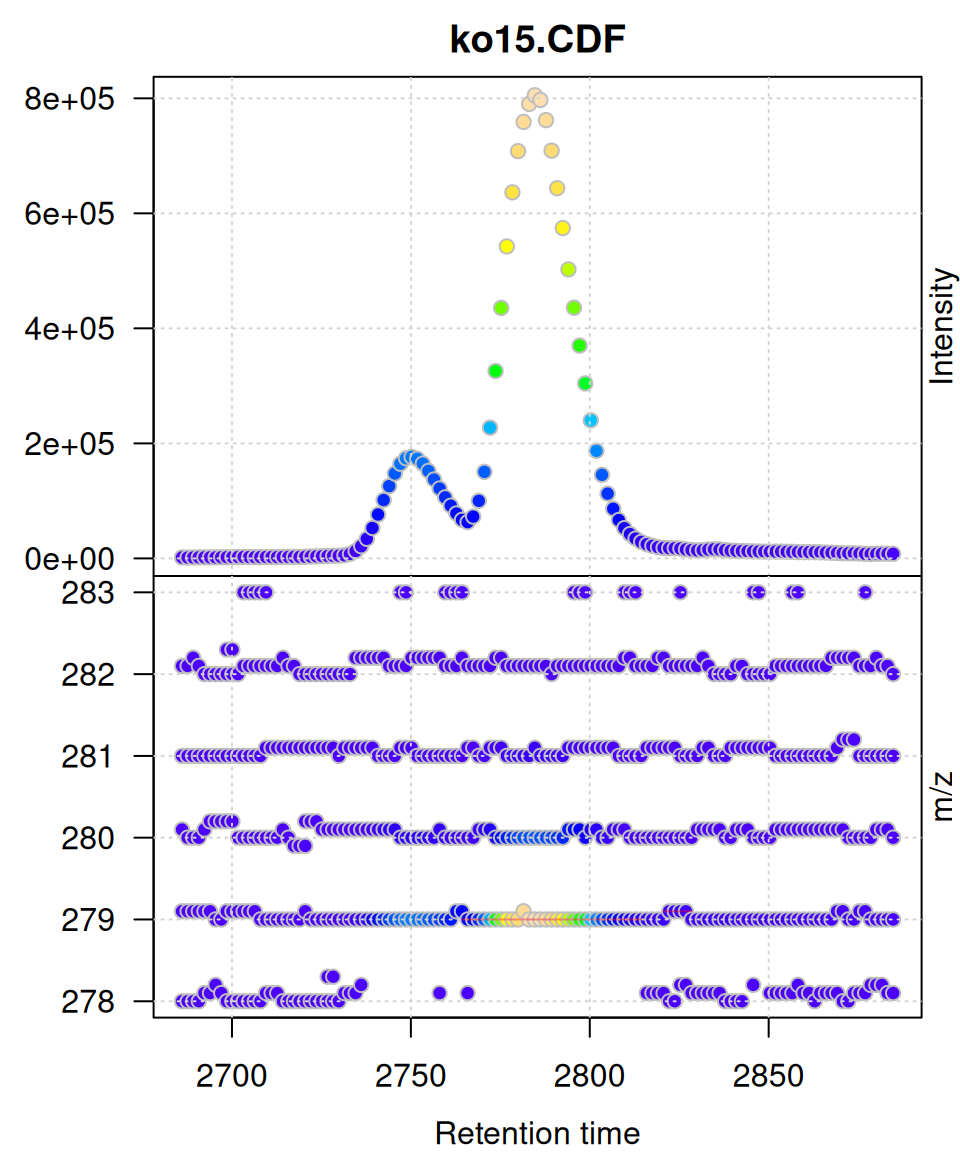
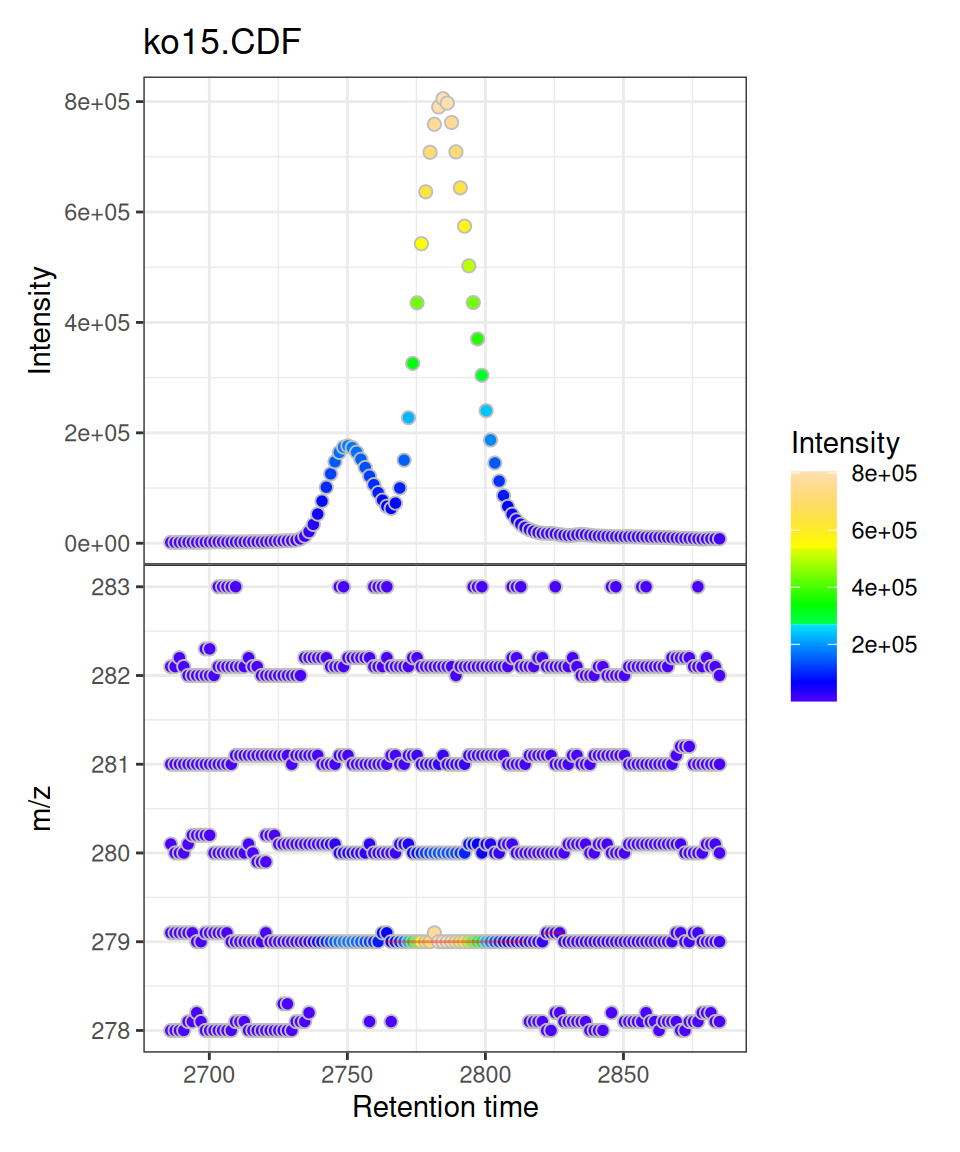
gplot(mse[1])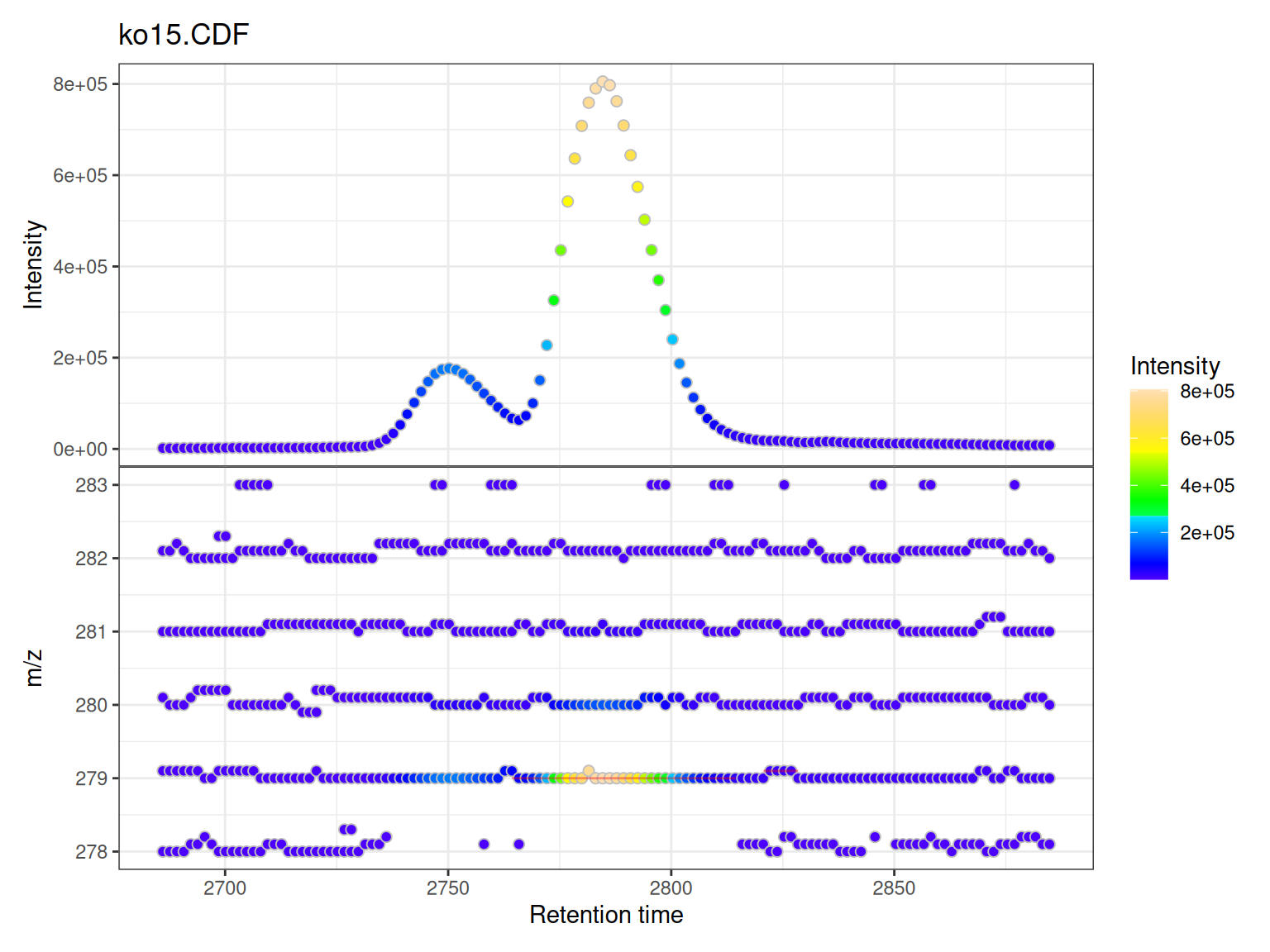
Understanding the Two Panels
Upper Panel: BPI Chromatogram
The Base Peak Intensity (BPI) shows the maximum intensity at each retention time across all m/z values in the filtered range.
- Each point represents one retention time
- Y-axis shows the maximum intensity observed at that time
- Color indicates intensity magnitude
- Useful for identifying retention time regions with strong signals
Lower Panel: m/z vs RT Scatter
The m/z vs retention time scatter shows the complete mass spectral data:
- X-axis: retention time
- Y-axis: m/z values
- Each point represents one data point (mass peak) from the raw spectra
- Color indicates intensity of that specific m/z at that retention time
- Shows the full two-dimensional structure of the LC-MS data
Peak Overlays
If chromatographic peaks have been detected (via findChromPeaks()), they are automatically overlaid as rectangles showing:
- Peak retention time boundaries (left/right edges)
- Peak m/z boundaries (top/bottom edges)
- Semi-transparent red color by default
Multiple Samples
When visualizing multiple samples, gplot() creates a vertically stacked layout:
# Plot all three samples
gplot(mse)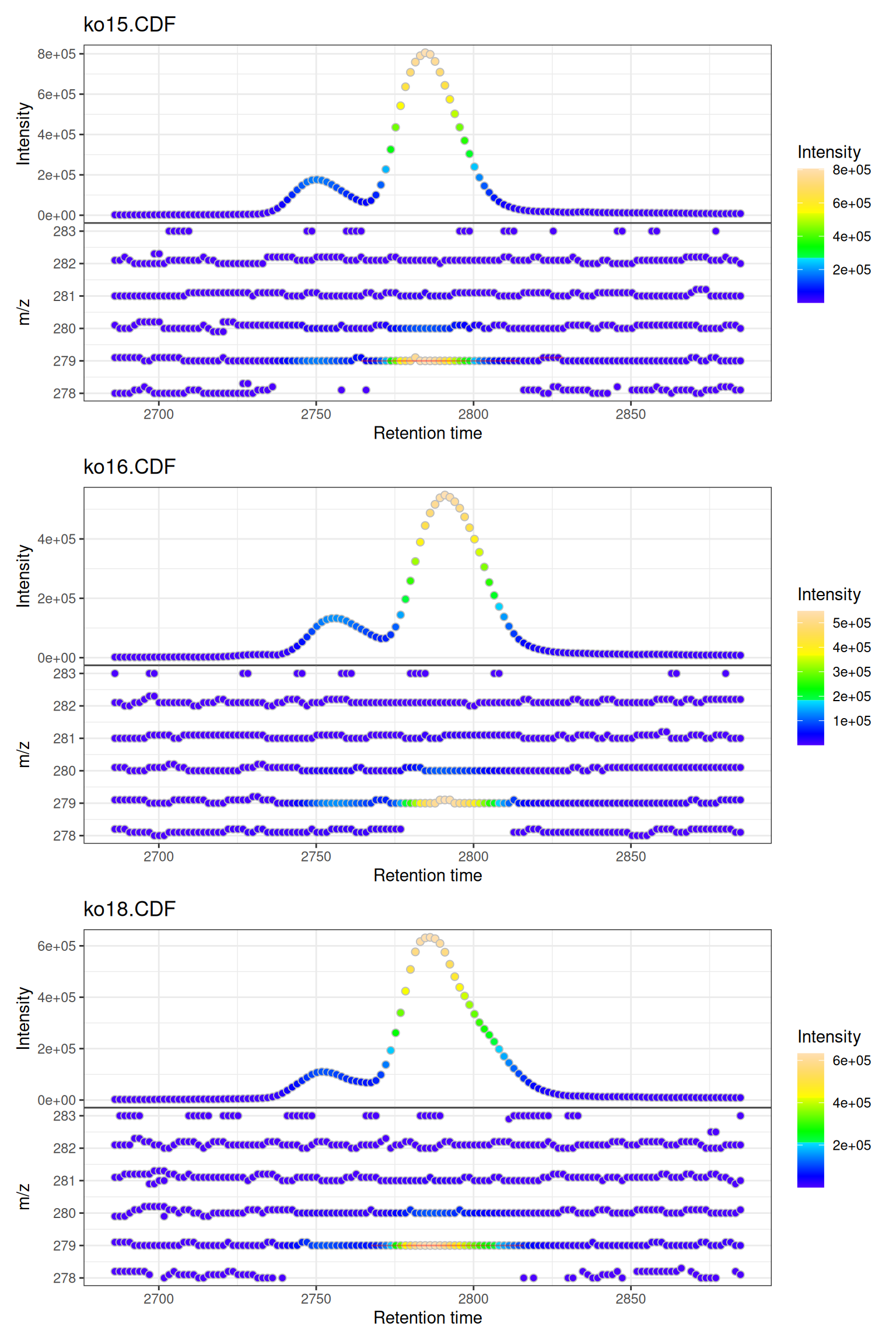
Customization
Custom Colors
gplot(mse[1],
col = "blue", # Point border color
peakCol = "red") # Peak rectangle color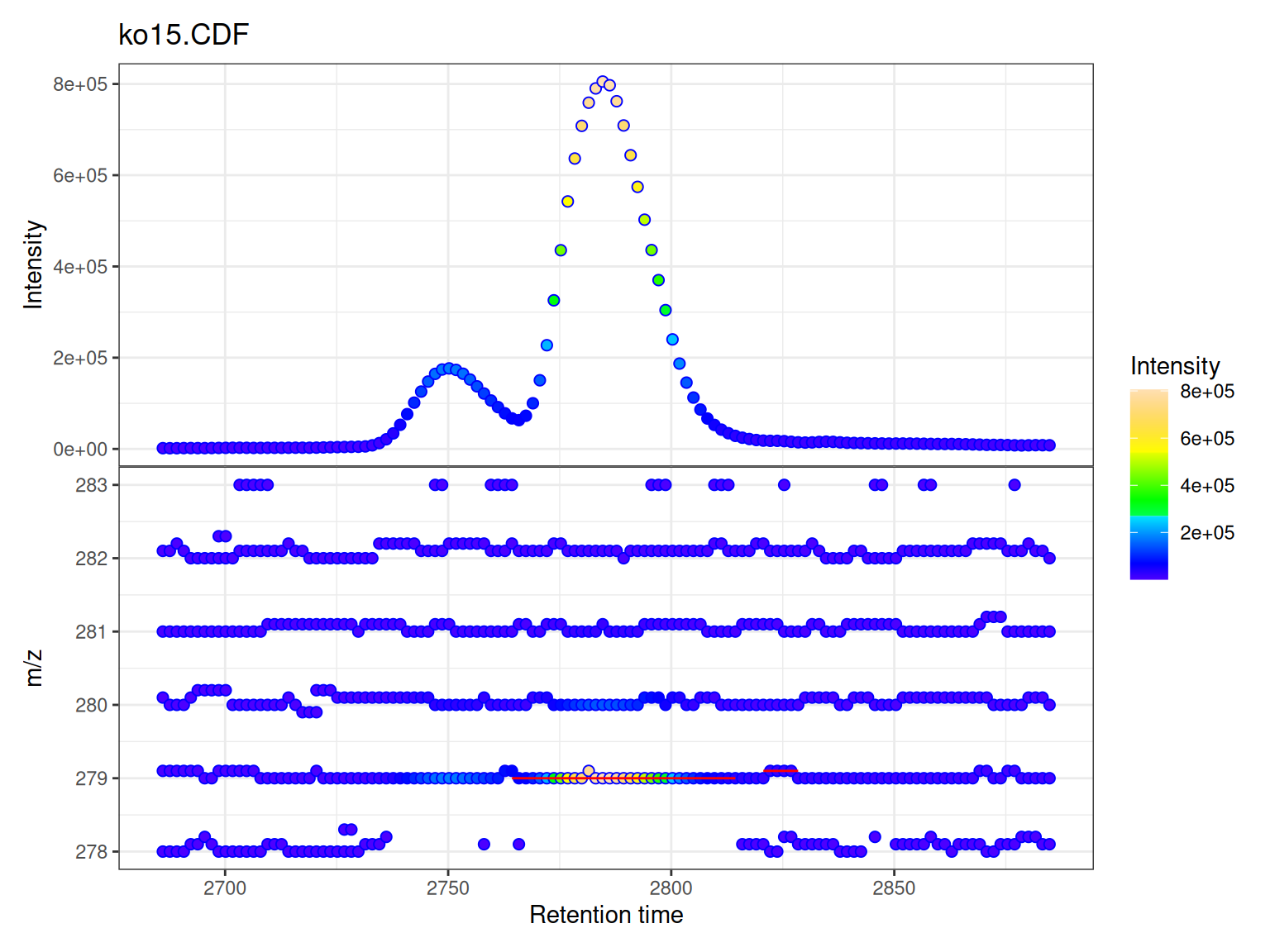
Custom Color Ramps
The intensity coloring uses a color ramp function. The viridis scales are reversed to follow MS convention (low=dark, high=bright):
library(viridisLite)
# Create reversed versions for MS convention (low=dark, high=bright)
viridis_rev <- function(n) rev(viridis(n))
magma_rev <- function(n) rev(magma(n))
plasma_rev <- function(n) rev(plasma(n))
p1 <- gplot(mse[1], colramp = viridis_rev) + ggtitle("Viridis")
p2 <- gplot(mse[1], colramp = magma_rev) + ggtitle("Magma")
p3 <- gplot(mse[1], colramp = plasma_rev) + ggtitle("Plasma")
(p1 | p2 | p3)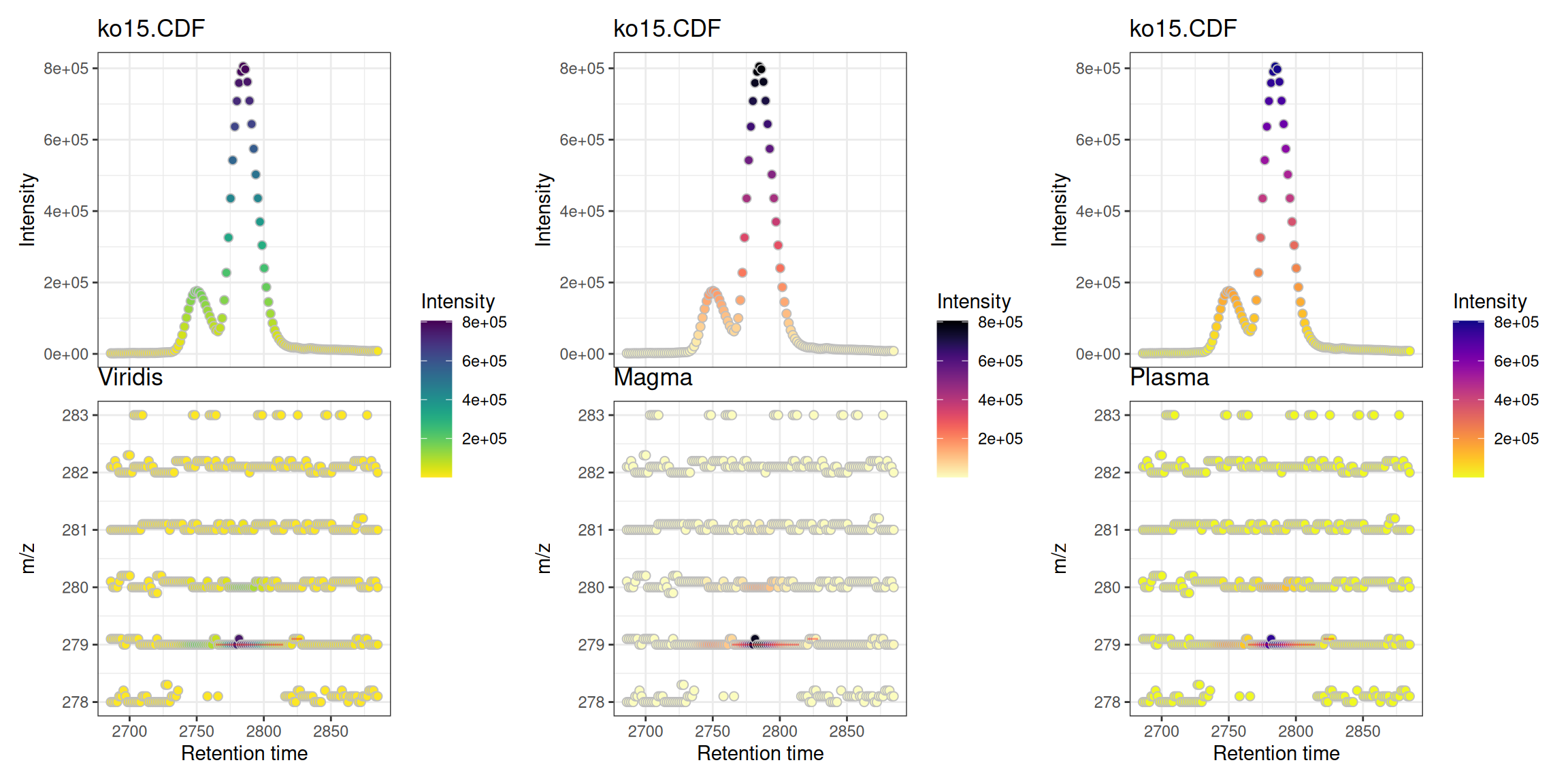
Custom Titles
# Plot shows sample names from data automatically
gplot(mse)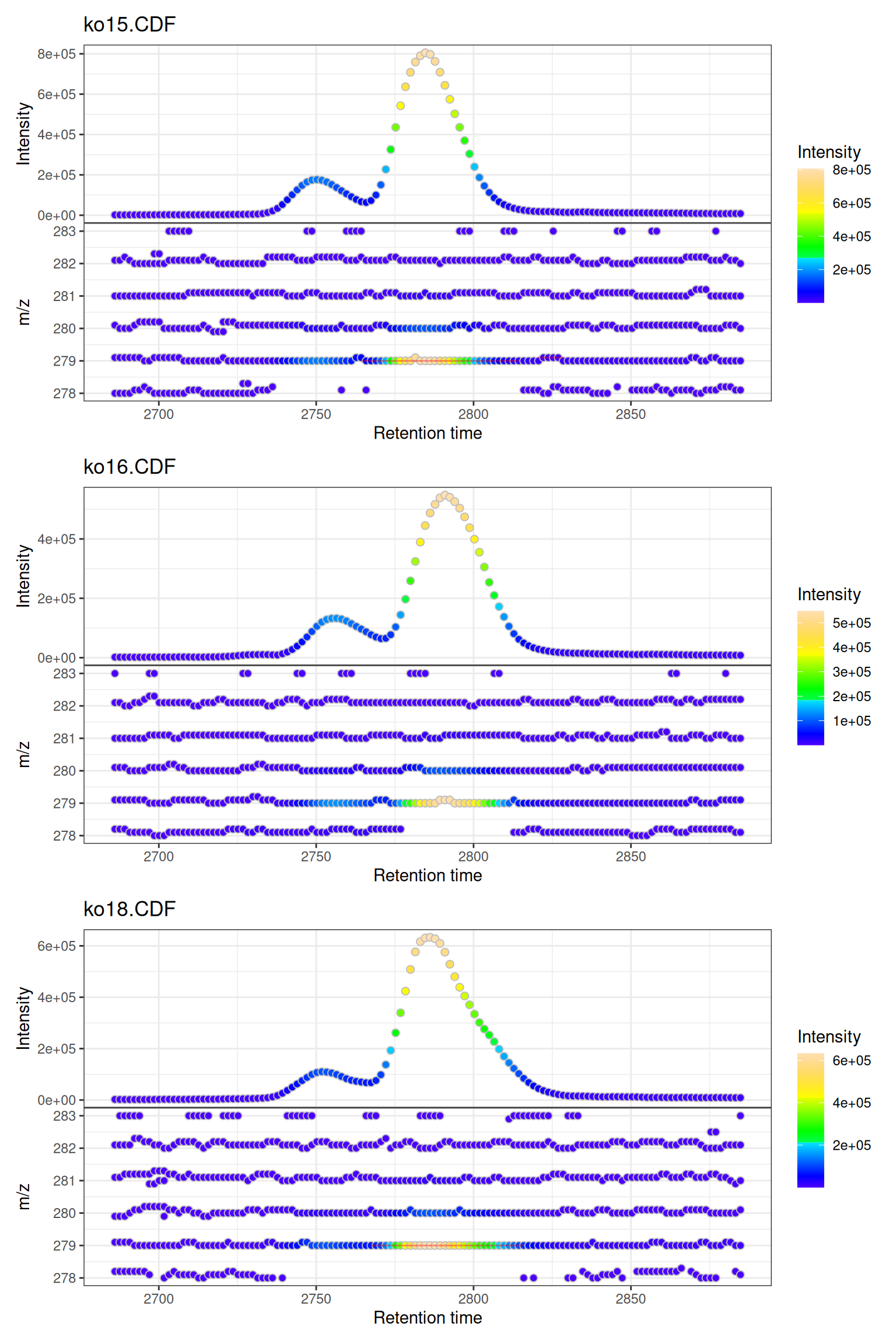
Interactive Visualization
Convert to interactive plotly for data exploration:
Accessing Individual Panels:
Since gplot() returns a patchwork object combining two panels, you can access and make each panel interactive separately:
This is useful when you want to customize each panel independently or embed them separately in reports.
Part 2: MS/MS Precursor Ion Visualization
Overview
The gplotPrecursorIons() function visualizes precursor ions selected for fragmentation in MS/MS (tandem mass spectrometry) experiments. This is particularly useful for:
- Assessing DDA (Data-Dependent Acquisition) performance
- Visualizing MS/MS coverage across the chromatographic run
- Quality control of MS/MS experiments
- Understanding which compounds were fragmented
What are Precursor Ions?
In MS/MS experiments, the mass spectrometer:
- Performs MS1 scans to detect all ions
- Selects specific ions (precursors) based on criteria (intensity, exclusion lists, etc.)
- Fragments these precursors and records MS2 spectra
The gplotPrecursorIons() function plots where and which precursor ions were selected across the LC-MS run.
Loading DDA Data
We’ll use example DDA data from the MsDataHub package:
# Load DDA MS/MS data
fl <- MsDataHub::PestMix1_DDA.mzML()
pest_dda <- readMsExperiment(fl)
# Check data structure
pest_dda
#> Object of class MsExperiment
#> Spectra: MS1 (4627) MS2 (2975)
#> Experiment data: 1 sample(s)
#> Sample data links:
#> - spectra: 1 sample(s) to 7602 element(s).Basic Precursor Ion Visualization
Default Plot
p <- gplotPrecursorIons(pest_dda)
p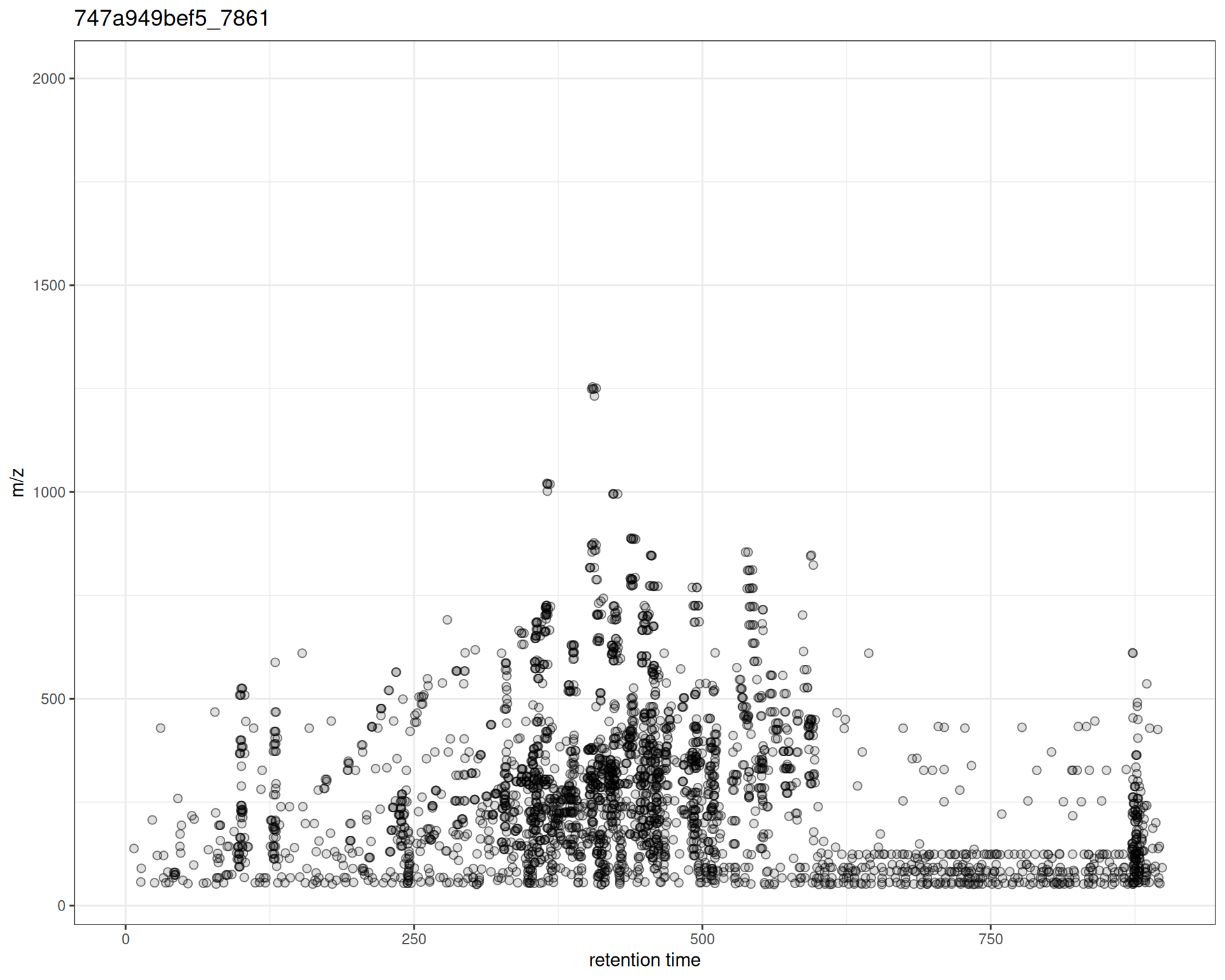
The plot shows:
- X-axis: Retention time when MS2 spectrum was acquired
- Y-axis: m/z of the precursor ion selected for fragmentation
- Points: Each precursor ion
Interpretation
From this plot, you can see:
- Distribution of MS/MS events across the chromatographic run
- m/z range of fragmented ions
- Density patterns - where fragmentation was most active
- Coverage gaps - RT or m/z regions without MS/MS data
Customization
Custom Colors and Symbols
p_custom <- gplotPrecursorIons(
pest_dda,
pch = 16, # filled circle
col = "#E41A1C" # point color
) +
labs(
x = "Retention Time (s)",
y = "Precursor m/z"
)
p_custom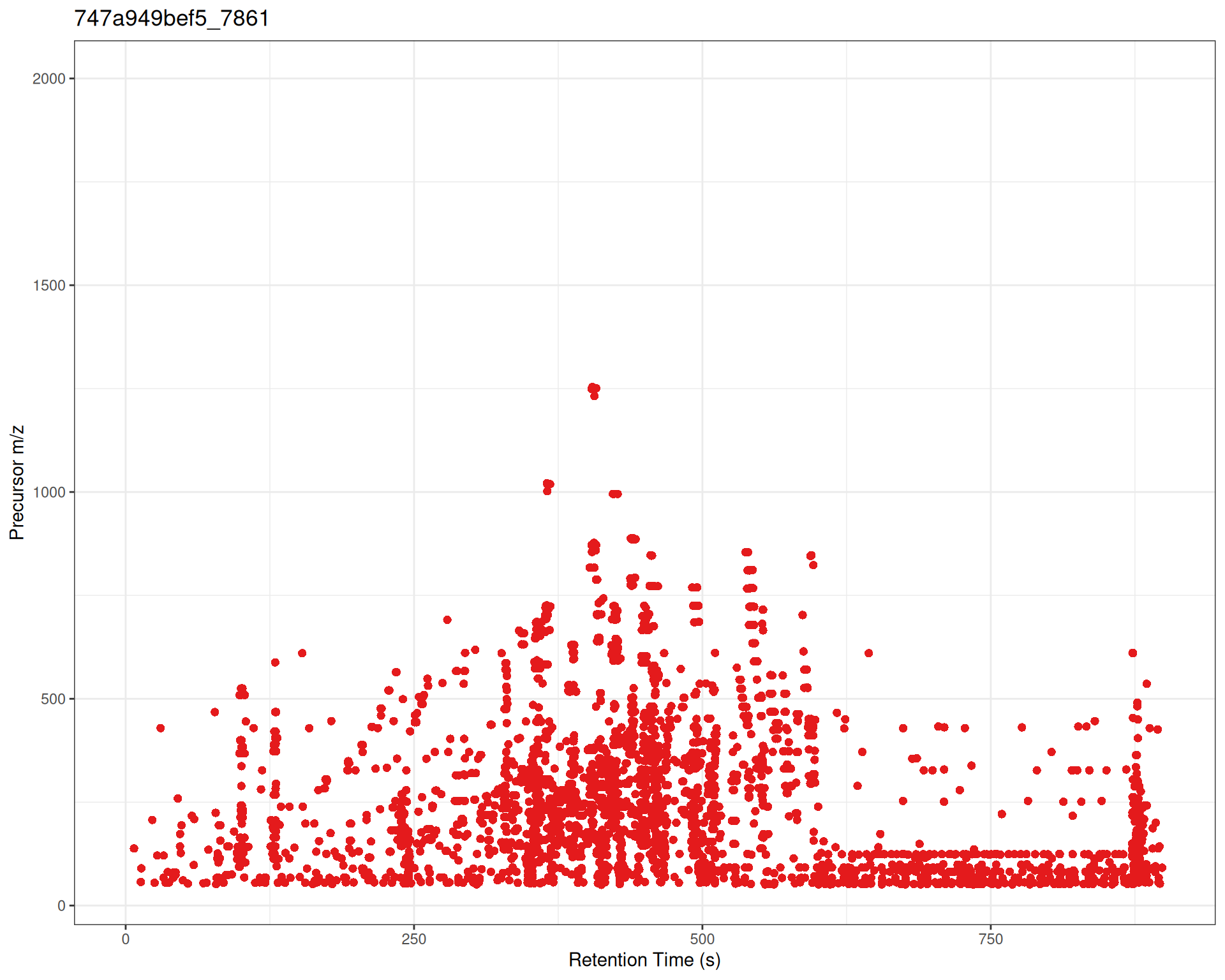
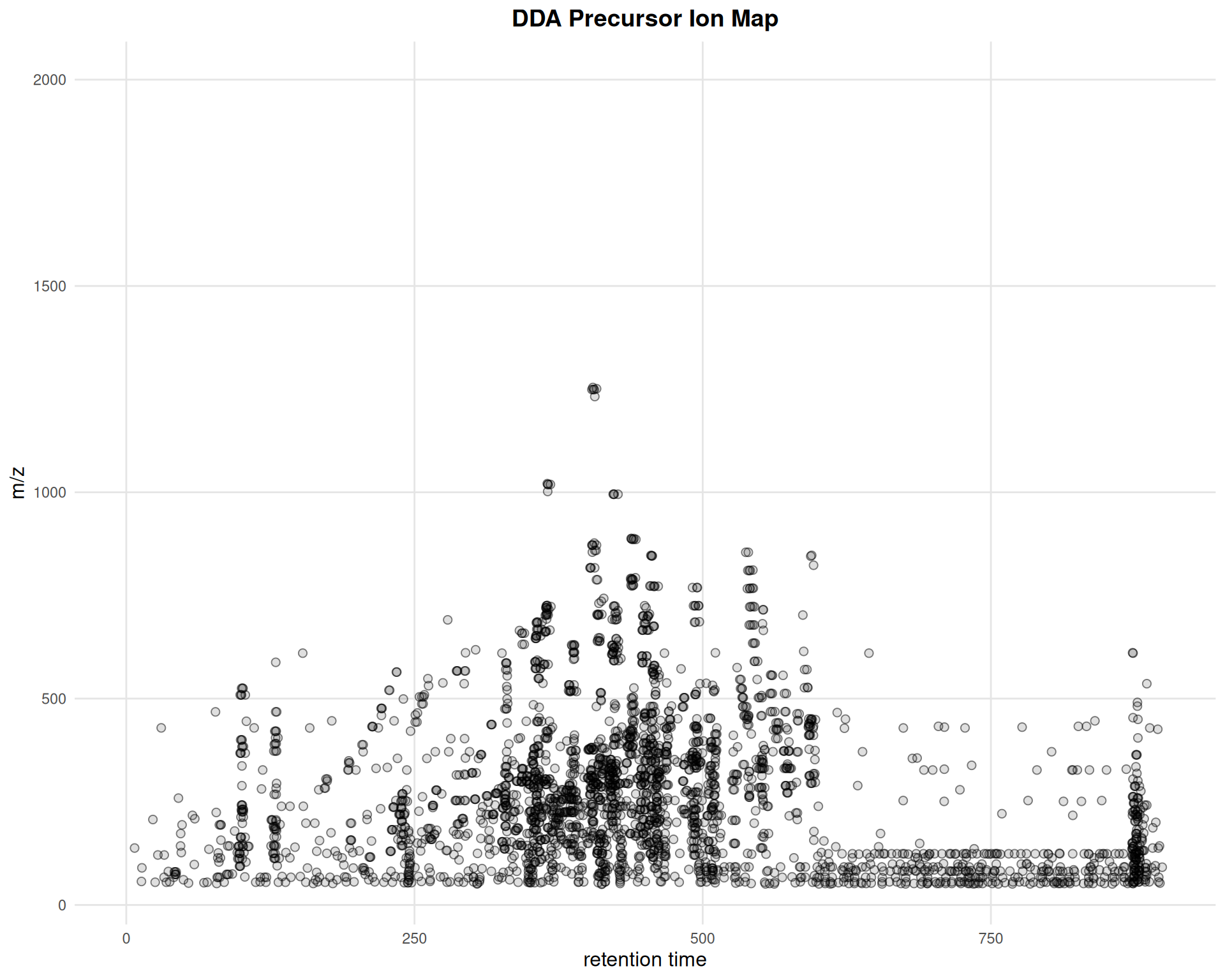
Adding ggplot2 Layers
gplotPrecursorIons(pest_dda) +
ggtitle("DDA Precursor Ion Map") +
theme_minimal() +
theme(
plot.title = element_text(hjust = 0.5, face = "bold", size = 14),
axis.title = element_text(size = 12),
panel.grid.major = element_line(color = "gray90"),
panel.grid.minor = element_blank()
)
Interactive Precursor Visualization
p_interactive <- gplotPrecursorIons(pest_dda)
ggplotly(p_interactive)Summary
When to Use
- Before any processing: Assess raw data quality
- Method development: Evaluate acquisition parameters
- Quality control: Ensure expected coverage and signals
- DDA experiments: Verify MS/MS performance
Next Steps
After visualizing and assessing your raw data, proceed to:
→ Step 2: Peak Detection - Detect chromatographic peaks in your data
Comparison with Original xcms
gplot(XcmsExperiment) vs plot()
gplotPrecursorIons() vs plotPrecursorIons()
Original xcms
plotPrecursorIons(pest_dda)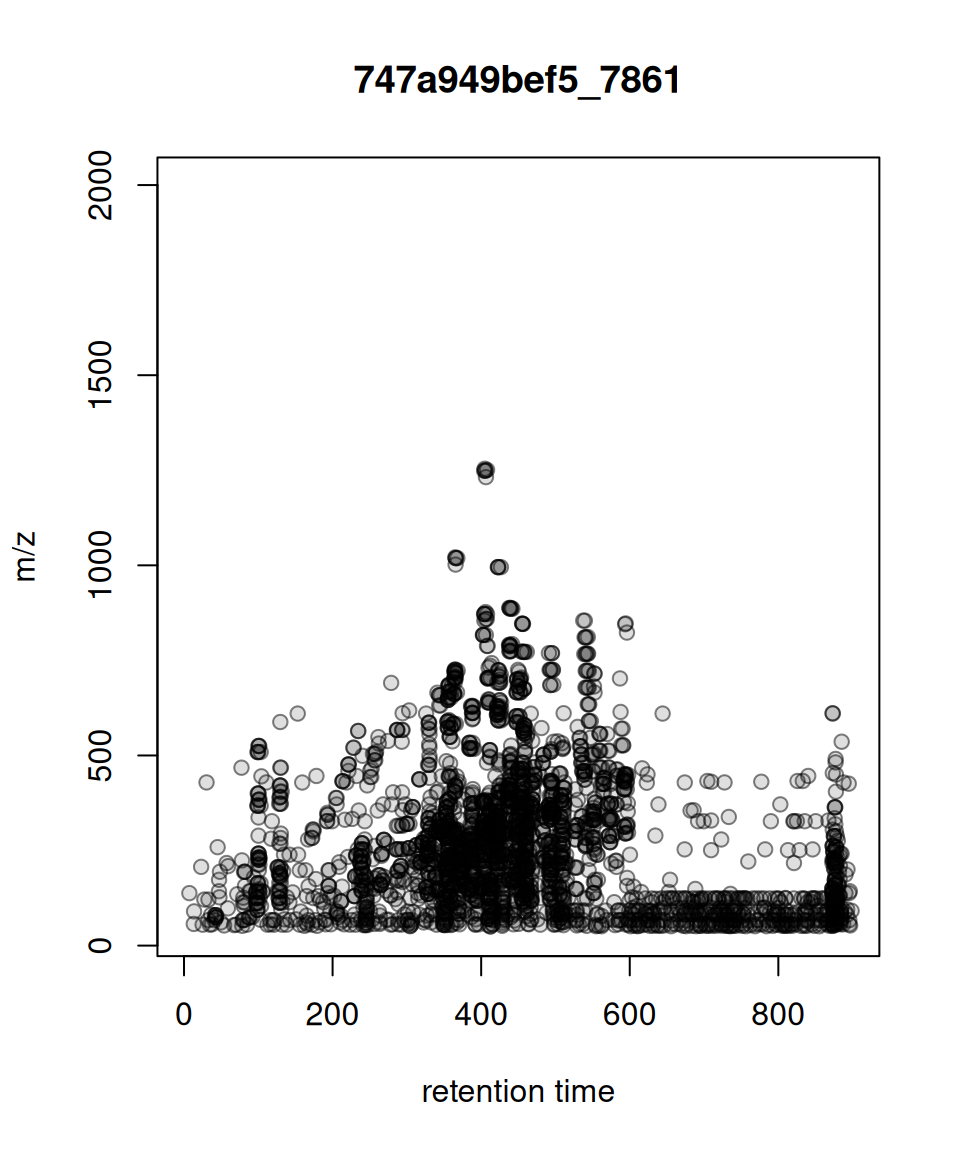
xcmsVis ggplot2
gplotPrecursorIons(pest_dda)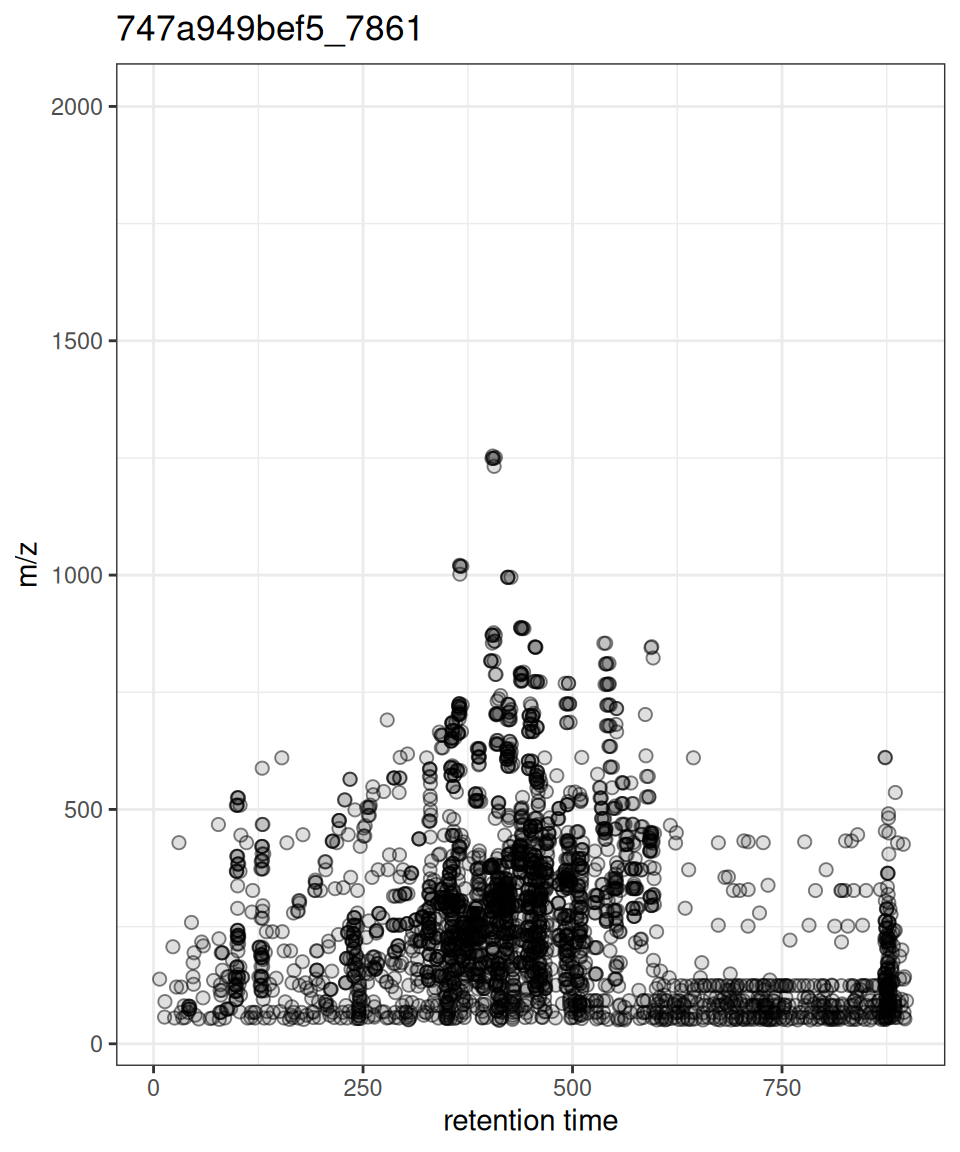
Session Info
sessionInfo()
#> R version 4.5.3 (2026-03-11)
#> Platform: x86_64-pc-linux-gnu
#> Running under: Ubuntu 24.04.3 LTS
#>
#> Matrix products: default
#> BLAS: /usr/lib/x86_64-linux-gnu/openblas-pthread/libblas.so.3
#> LAPACK: /usr/lib/x86_64-linux-gnu/openblas-pthread/libopenblasp-r0.3.26.so; LAPACK version 3.12.0
#>
#> locale:
#> [1] LC_CTYPE=C.UTF-8 LC_NUMERIC=C LC_TIME=C.UTF-8
#> [4] LC_COLLATE=C.UTF-8 LC_MONETARY=C.UTF-8 LC_MESSAGES=C.UTF-8
#> [7] LC_PAPER=C.UTF-8 LC_NAME=C LC_ADDRESS=C
#> [10] LC_TELEPHONE=C LC_MEASUREMENT=C.UTF-8 LC_IDENTIFICATION=C
#>
#> time zone: UTC
#> tzcode source: system (glibc)
#>
#> attached base packages:
#> [1] stats graphics grDevices utils datasets methods base
#>
#> other attached packages:
#> [1] viridisLite_0.4.3 MsDataHub_1.10.0 patchwork_1.3.2
#> [4] plotly_4.12.0 ggplot2_4.0.2 MsExperiment_1.12.0
#> [7] ProtGenerics_1.42.0 xcmsVis_0.99.10 xcms_4.8.0
#> [10] BiocParallel_1.44.0 BiocStyle_2.38.0
#>
#> loaded via a namespace (and not attached):
#> [1] RColorBrewer_1.1-3 jsonlite_2.0.0
#> [3] MultiAssayExperiment_1.36.1 magrittr_2.0.4
#> [5] farver_2.1.2 MALDIquant_1.22.3
#> [7] rmarkdown_2.30 fs_1.6.7
#> [9] vctrs_0.7.1 memoise_2.0.1
#> [11] htmltools_0.5.9 S4Arrays_1.10.1
#> [13] BiocBaseUtils_1.12.0 progress_1.2.3
#> [15] AnnotationHub_4.0.0 curl_7.0.0
#> [17] SparseArray_1.10.9 mzID_1.48.0
#> [19] htmlwidgets_1.6.4 plyr_1.8.9
#> [21] httr2_1.2.2 impute_1.84.0
#> [23] cachem_1.1.0 igraph_2.2.2
#> [25] lifecycle_1.0.5 iterators_1.0.14
#> [27] pkgconfig_2.0.3 Matrix_1.7-4
#> [29] R6_2.6.1 fastmap_1.2.0
#> [31] MatrixGenerics_1.22.0 clue_0.3-67
#> [33] digest_0.6.39 pcaMethods_2.2.0
#> [35] AnnotationDbi_1.72.0 S4Vectors_0.48.0
#> [37] ExperimentHub_3.0.0 crosstalk_1.2.2
#> [39] GenomicRanges_1.62.1 RSQLite_2.4.6
#> [41] filelock_1.0.3 Spectra_1.20.1
#> [43] labeling_0.4.3 httr_1.4.8
#> [45] abind_1.4-8 compiler_4.5.3
#> [47] bit64_4.6.0-1 withr_3.0.2
#> [49] doParallel_1.0.17 S7_0.2.1
#> [51] DBI_1.3.0 MASS_7.3-65
#> [53] rappdirs_0.3.4 DelayedArray_0.36.0
#> [55] mzR_2.44.0 tools_4.5.3
#> [57] PSMatch_1.14.0 otel_0.2.0
#> [59] glue_1.8.0 QFeatures_1.20.0
#> [61] grid_4.5.3 cluster_2.1.8.2
#> [63] reshape2_1.4.5 generics_0.1.4
#> [65] gtable_0.3.6 preprocessCore_1.72.0
#> [67] tidyr_1.3.2 data.table_1.18.2.1
#> [69] hms_1.1.4 MetaboCoreUtils_1.18.1
#> [71] XVector_0.50.0 BiocGenerics_0.56.0
#> [73] BiocVersion_3.22.0 foreach_1.5.2
#> [75] pillar_1.11.1 stringr_1.6.0
#> [77] limma_3.66.0 dplyr_1.2.0
#> [79] BiocFileCache_3.0.0 lattice_0.22-9
#> [81] bit_4.6.0 tidyselect_1.2.1
#> [83] Biostrings_2.78.0 knitr_1.51
#> [85] IRanges_2.44.0 Seqinfo_1.0.0
#> [87] SummarizedExperiment_1.40.0 stats4_4.5.3
#> [89] xfun_0.56 Biobase_2.70.0
#> [91] statmod_1.5.1 MSnbase_2.36.0
#> [93] matrixStats_1.5.0 stringi_1.8.7
#> [95] lazyeval_0.2.2 yaml_2.3.12
#> [97] evaluate_1.0.5 codetools_0.2-20
#> [99] MsCoreUtils_1.22.1 tibble_3.3.1
#> [101] BiocManager_1.30.27 cli_3.6.5
#> [103] affyio_1.80.0 Rcpp_1.1.1
#> [105] MassSpecWavelet_1.76.0 dbplyr_2.5.2
#> [107] png_0.1-9 XML_3.99-0.22
#> [109] parallel_4.5.3 blob_1.3.0
#> [111] prettyunits_1.2.0 AnnotationFilter_1.34.0
#> [113] MsFeatures_1.18.0 scales_1.4.0
#> [115] affy_1.88.0 ncdf4_1.24
#> [117] purrr_1.2.1 crayon_1.5.3
#> [119] rlang_1.1.7 vsn_3.78.1
#> [121] KEGGREST_1.50.0